
Frontline workers at Ganado receive vaccine
Navajo Times | Donovan Quintero
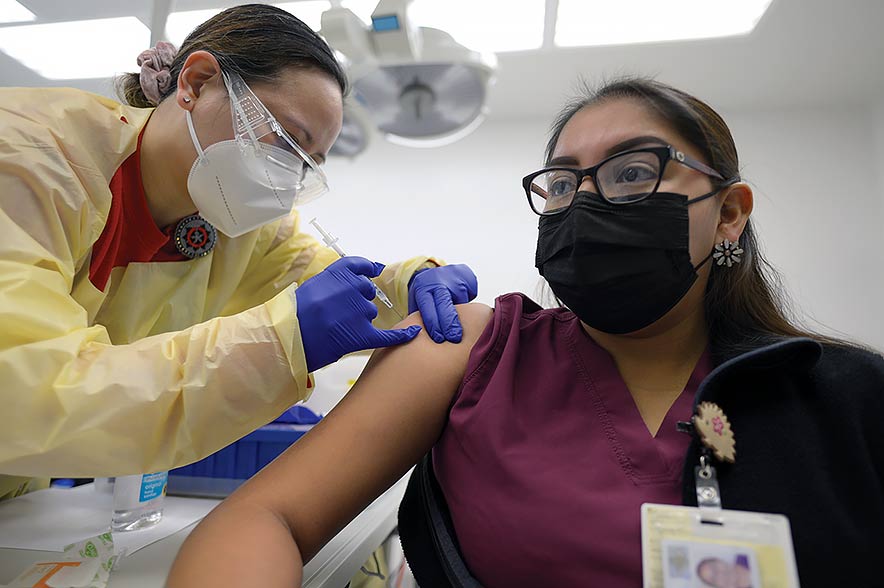
A health care worker receives one of the first doses of the Pfizer/BioNtech vaccine to arrive on the reservation. the Indian Health Service is expecting another batch of vaccines, this time the Moderna vaccine, next week.
GANADO
Shaiyaia Fowler, a medical assistant at Sage Memorial Hospital, rolled up her sleeves on Tuesday morning to become part of history. She didn’t wince when the hypodermic needle penetrated her skin to inject the Pfizer BioNTech COVID-19 vaccine.
The vaccine was authorized for emergency use under the Emergency Use Authorization Act last weekend.
“As long as I am protecting myself, my kids and family, I feel fine,” Fowler said after she was given the shot.
Fowler, who works with COVID-19 patients in the Outpatient Clinic, said her children are eight, three, and three months old. Fowler stressed the importance of continuing to wear a mask, which, to her, protects infants and the elderly from getting the respiratory disease.
Escorted by Navajo Nation Police officers, the first shipment of the vaccine for health care workers, long-term nursing patients and staff arrived on Monday. The drug, which is required to be stored at -90 degrees Fahrenheit, was carried in by Cmdr.
Erica Harker with the U.S. Public Health Service, in an ice chest. The vaccine would be used for 130 of the Public Law 93-638 hospital staff who signed up and agreed to take it.
Sage Memorial Hospital serves approximately 23,000 residents of Ganado, Kinlichee, Klagetoh, Wide Ruins, Lower Greasewood Springs, Cornfields, Nazlini and Steamboat, according to the hospital’s Incident Commander Chris West.
All staff who agreed to take vaccine work directly with patients infected with the highly contagious virus, West said. A total of 3,900 doses of the vaccine was delivered to health care facilities across the reservation.
Navajo Area Indian Health Service spokeswoman Genevieve Notah said the Crownpoint Service unit vaccinated a few employees on Monday afternoon and will continue vaccinating frontline workers and other first responders throughout the week. “Other service units will be vaccinating this week too,” she said on Monday.
Sage hospital received 33 vials, or 165 doses, that would be used to vaccinate their frontline workers, according to hospital spokesman Gary Pahe. West added the hospital is expecting to receive the Moderna vaccine within the next seven to 10 days. The Moderna vaccine was also authorized by the FDA under the EUA. According to the company that made the drug, the two-dose vaccine was 94% effective.
After getting her shot, Fowler, a Steamboat, Arizona, native, joined her colleagues in a separate room where they would be observed for any “adverse reaction,” which West said may include headaches, fever and body aches.
As of Tuesday, the Navajo Nation Department of Health reported 19,929 Navajo people have contracted the virus and 727 people have died. To help the fight against the virus, the FDA also approved the emergency use of an at-home COVID-19 test that can be administered without a health care worker. People can obtain a sample themselves with an over-the-counter nasal swab test kit, the FDA stated.
Users of the $30 self-testing kit would be required to download an app that provides them step-by-step instructions. The results can be shared with employees, schools and health care professionals. West wanted to emphasize the Pfizer vaccine is not a cure and people who were vaccinated could still contract the virus.
“It’s still possible to get it,” he said. “So everybody needs to continue to practice the social distancing, wearing their mask when they’re out in public and washing their hands.”
Anyone who begins to experience any symptoms should get tested, West said. “Everybody just needs to monitor themselves for any symptoms,” West said. “Once they start feeling any kind of symptoms, as far as fever, cough, and a headache that’s lasting for a long period, runny nose — diarrhea, if they’re experiencing these symptoms, they should go into their closest emergency room and be seen.”
As a public service, the Navajo Times is making all coverage of the coronavirus pandemic fully available on its website. Please support the Times by subscribing.
How to protect yourself and others.
Why masks work. Which masks are best.
Resources for coronavirus assistance








 Highway 264,
Highway 264, I-40, WB @ Winslow
I-40, WB @ Winslow